
Rechargeable batteries store energy in the form of chemical energy and convert the chemical energy into electrical energy for use by our devices during discharge. In a rechargeable battery, chemical energy is converted into electrical energy during discharge and electrical energy is converted into chemical energy during charge. Electrical energy typically refers to electrons flowing through a wire, while chemical energy is the energy stored in the bonds of atoms and molecules.

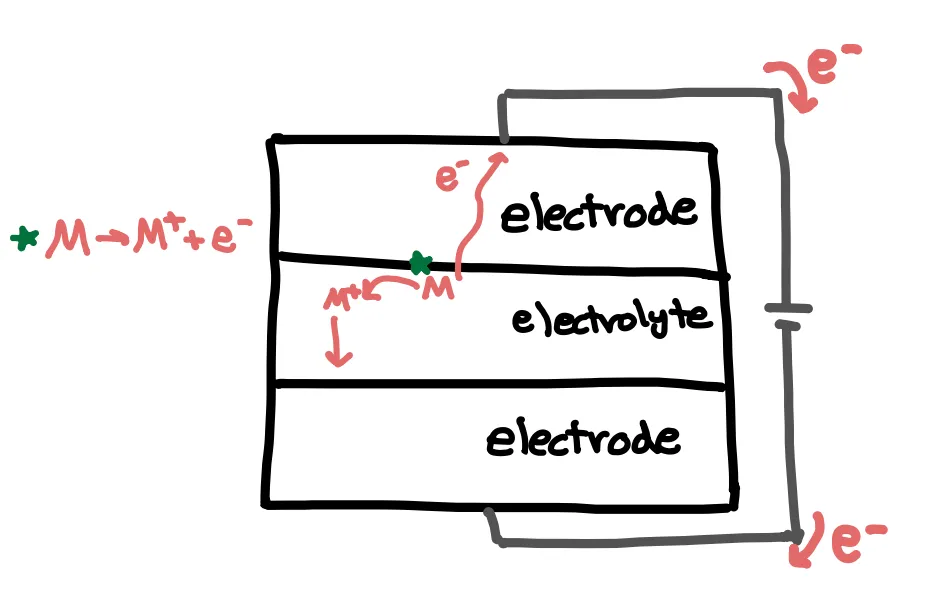
A typical battery consists of two electrodes with a potential difference across them. Electrons want to flow from a higher energy state to a lower energy state, similar to how a ball wants to roll downhill. If we directly put the electrodes in contact with each other, electrons spontaneously transfer, and we can’t harness any energy from this reaction. By physically separating the electrodes and connecting the electrodes with a wire, the electrons instead flow through the wire, going on to power your device. When we want to recharge the battery, we can reverse the process, similar to how we could carry a ball up the hill again.
But where do these electrons come from? This is where the chemistry comes into play. Let’s call our element or compound M. At the lower potential electrode, commonly called the anode, . The electron flows through the external circuit, while the resulting ion migrates through the electrolyte to the higher potential electrode, commonly called the cathode. Here, the ion gains an electron from the external circuit to convert back to ().
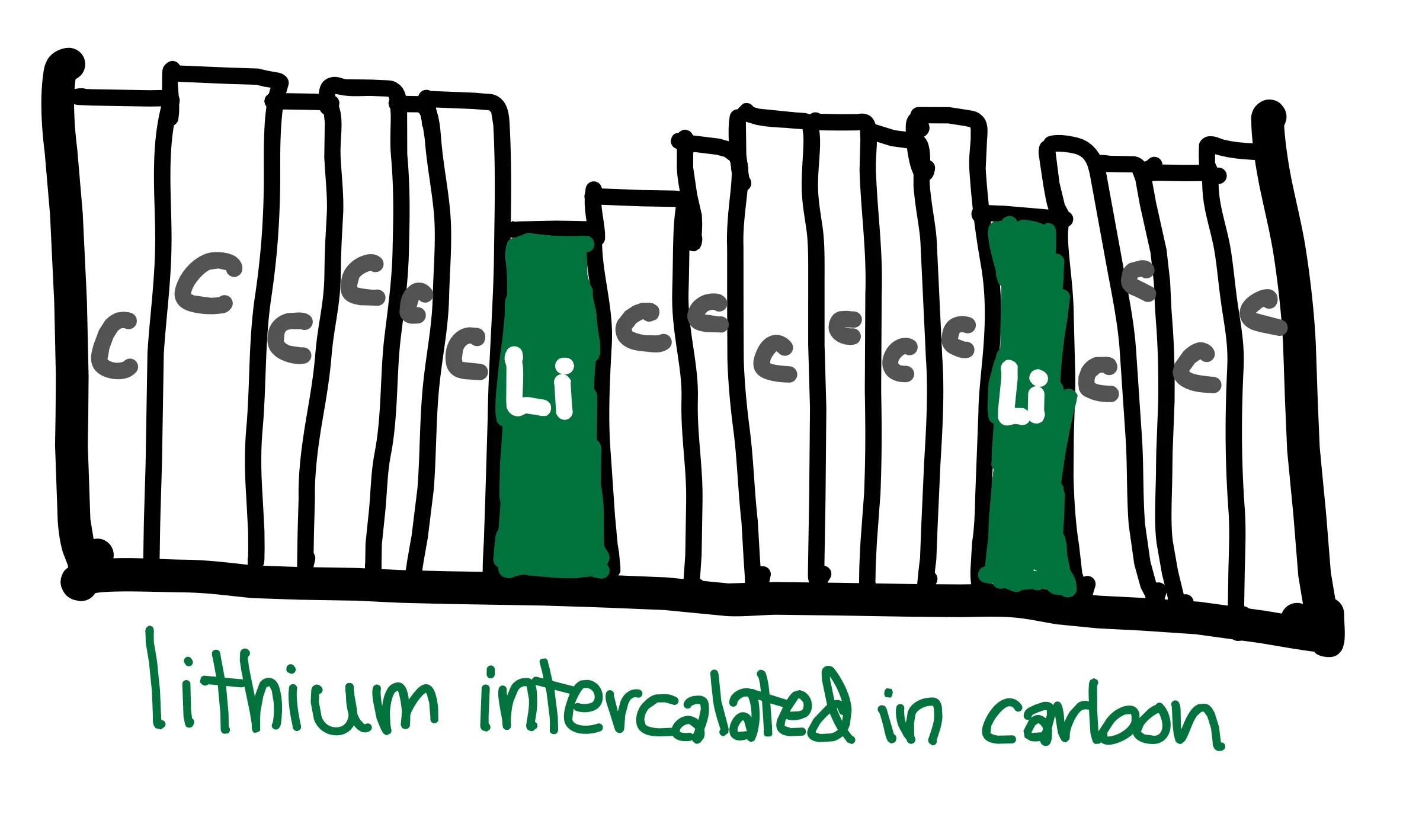
That might sound and look a little obscure … let’s look at a real system of a lithium-ion battery. Most rechargeable batteries that we use are lithium-ion batteries – think phones, computers, and electric vehicles. Since they were first commercialized in 1991, many different Li-ion battery variations have been created. Generally, a lithium-ion battery has a carbon anode and a transition metal cathode. Lithium ions slide into layers of the electrodes like books slide into a bookshelf. The processes of charging and discharging is illustrated below, with a cobalt oxide cathode. It is key that there are two half cell reactions that are physically separated, one at each electrode, in order to allow the overall reaction to occur while moving electrons through an external circuit.

Battery systems differ in the functions that the components have, but generally reactions occur at the electrode-electrolyte interface. The electrolyte allows for the transfer of ions between electrodes, and a separator physically separate the electrodes. I discuss the components of a battery in more detail in the next post.